DYD solution
Design Your Diary

Designed for You,
Tailored to Your Needs
Our e-diary solution adapts to your workflow with unmatched flexibility. Fully customizable notifications can be triggered by any action or scheduled as periodic reports. Advanced data visualization lets you define who sees what, ensuring precise access control and meaningful insights.
Compliant with FDA and EU regulations, including 21 CFR Part 11, our secure, out-of-the-box solution simplifies clinical data management. With robust audit trails and intuitive query management, data integrity is guaranteed—all at an economical cost, delivering efficiency without compromise.
DYD Values
Independence
Fostering self-reliance, reducing unnecessary third party dependencies.
Efficiency
Streamlining processes to enhance trial management and overall efficiency.
Transparency
Commitment to clear, open communication, ensuring trust in every interaction.
Cost Efficiency
Delivering economic tools to significantly reduce trial costs without compromising quality.
Regulatory Excellence
Ensuring regulatory compliance by default, providing a solid foundation for seamless trials.
Continuous Evolution
Offering a live product that evolves organically, integrating client needs and providing ongoing assistance.
DYD In Action
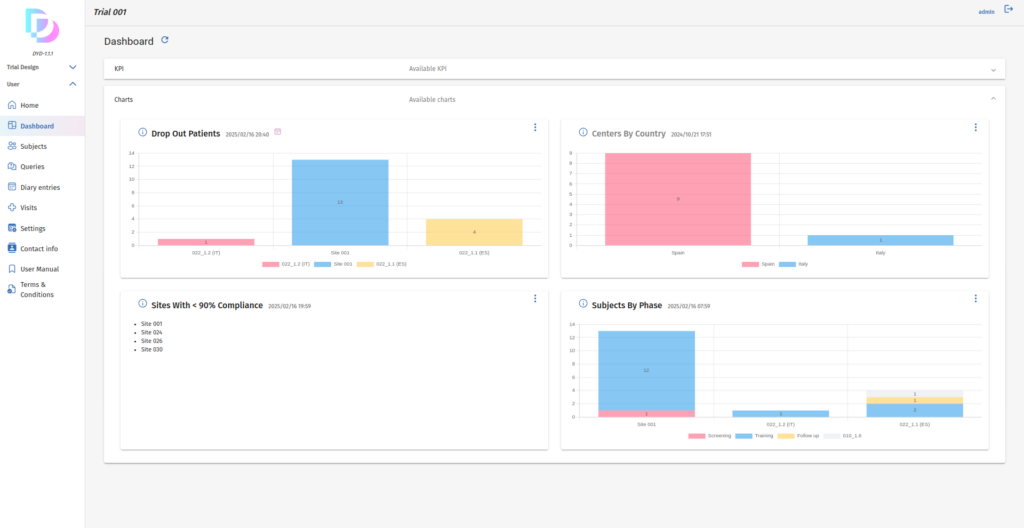
Customizable Dashboard
Instantly generate KPIs and charts with our intuitive dashboard builder.
Define visibility settings to control who sees what, ensuring tailored insights for each user while maintaining data security and relevance.

Access on Any Device
Access your e-diary seamlessly from any device—smartphone, tablet, or desktop.
Our BYOD-compatible, multiplatform solution ensures flexibility and accessibility, allowing users to engage effortlessly while maintaining security and compliance across all operating systems.
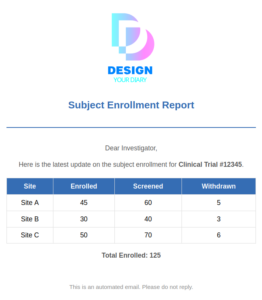
Seamless Notification & Report
Set up dynamic notifications and alerts at any point during the trial.
Include real-time updates, summary reports, or any critical data—ensuring timely communication and complete flexibility in managing study events.
e-Diary Specifications
DYD seamlessly generates e-diary specifications based on your configuration.
Adjust settings anytime during the trial, and a new version is instantly created.
This ensures accuracy, flexibility, and up-to-date documentation throughout the study.
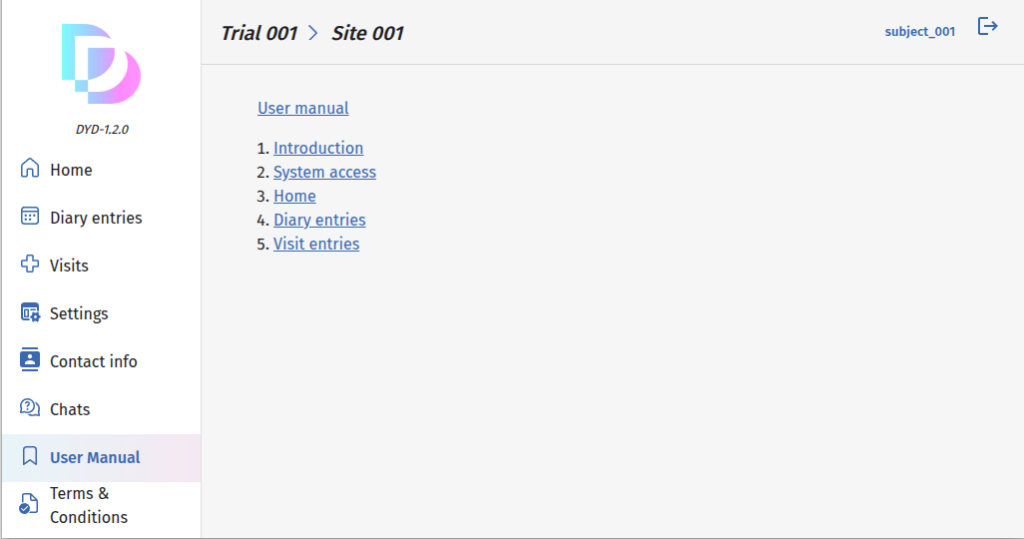
Dynamic Embedded User Manual
Our embedded, dynamic user manual adapts to each user’s access level, displaying only relevant sections.
Seamlessly integrated within the platform, it provides real-time guidance, ensuring an intuitive and efficient experience tailored to individual roles and permissions.
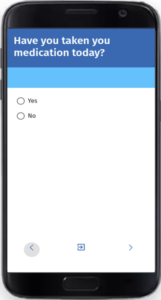
Effortless, Visual Questionnaire Design
Easily create highly configurable questionnaires with our user-friendly, visual interface.
Designed for efficiency, our intuitive tool empowers users to design complex forms effortlessly, ensuring precision and adaptability without requiring technical expertise.

Integrated Text & Video Chat for Seamless Collaboration
Enable seamless interaction through integrated text and video chat.
Fully configurable role-based permissions ensure controlled access, while help desk users can provide real-time assistance—enhancing collaboration and support across the platform.

Full Control
Self-Managed or Expert-Assisted Setup
Choose full autonomy or expert guidance—configure your e-diary independently or receive our support at any stage.
Our flexible approach ensures you have complete control while assistance is always available if needed.
DYD Key Features
Your Needs, Our Commitment
We prioritize your goals by delivering innovative, compliant, and user-friendly solutions.
Our e-diary ensures seamless data collection, security, and efficiency, empowering you with full control while exceeding industry standards.
Your success drives our dedication to excellence.
Audit Trail
Ensures complete traceability with secure, time-stamped records of all data modifications.
Query Management
Streamlines issue resolution with real-time data validation and query tracking.
Data Transfer
Securely exports and integrates clinical data with external systems for seamless interoperability.
Custom Notifications
Delivers real-time and periodic notifications tailored to user roles and study requirements.
Data Visualization
Provides highly customizable charts and dashboards for clear insights and trend analysis.
Data Capture
Enables accurate, compliant, and user-friendly electronic data collection even when user is offline.